Oxygenates
The primary objective of any race engine is to produce power – preferably lots of it – but to do so a number of factors need to be in place. Clearly it needs a plentiful supply of oxygen, so the engine should be designed to gulp as much air as possible and retain it inside the cylinder. However, that is not the only supply of oxygen available, for lurking inside most gasoline fuels these days is another source, and although it is ‘locked’ up in the fuel in the form of a liquid, it has just as much potential to burn and therefore produce power as any other form of the chemical. To fuel chemists and combustion engineers, this extra supply is often referred to as ‘oxygenates’ and will be blended into gasoline fuels to create the optimum combustion characteristics commensurate with the requirements of the engine and, if relevant, environmental/regulatory considerations.
Although there are many types of oxygenate, only two types are commonly used – the alcohol family of methanol, ethanol, iso-propanol or TBA (tertiary butanol); and the ether family of MTBE (methyl tertiary butyl ether) or ETBE (ethyl tertiary butyl ether).
In terms of the relative oxygen content in the fuel molecule, alcohols tend to contain the most, with methanol the greatest at around 50% by weight. The more oxygen in an alcohol when blended with gasoline, the less in theory that has to be drawn in through the engine intake valve, and I suppose therefore in terms of an outright performance increase, methanol should be top of the list. And so you might expect, but for the fact that the lower heating value of methanol is less than half that of your normal gasoline, so what you add in terms of oxygen content you take away in the form of heating value, and to burn all the intake air from your typical engine it will take around twice as much as that of your usual gasoline it replaces.
The great advantages of the alcohol fuels like methanol, however, are their high heat of evaporation (six times that of regular gasoline) and their exceptional resistance to combustion knock, which make them ideal for supercharged or turbocharged engines. On the negative side, methanol has poor solubility in gasoline, which is partly why ethanol, the higher alcohol, is more readily blended at the pumps – this and perhaps the reason that ethanol can be manufactured by fermentation of biomass, which makes it much more politically expedient in the modern environmentally (and politically) aware world. The blending of alcohol fuels, particularly methanol into gasoline, can also have a significant effect on the vapour pressure of the resultant fuel, especially at low treat rates [see Figs. 1 and 2]. This can make it a challenge to blend a fuel to within the narrow band of vapour pressures necessary to give consistent combustion.
Of the other oxygenates available, until ten to 15 years ago, MTBE was the most common blend component for gasoline. Containing a useful 18% oxygen by weight, its heating value is only slightly less than that of the gasoline with which it is mixed. The chief attractions of MBTE, however, are its high level of knock resistance (around 115-123 RON) and its benign effect on the fuel vapour pressure of the resulting gasoline blend, particularly at low treat rates. Added to some fuels by as much as 15%, political pressure in the US led to the banning of MTBE in fuels because of ground water contamination issues in bulk storage tanks, and was replaced by ethanol. Nevertheless, many race fuels still use MTBE which, when allowed, is far less corrosive to an engine’s internal parts than alcohol.
They may not be as environmentally acceptable as ethanol, but for naturally aspirated engines the lower ethers such as MBTE have much to recommend themselves.
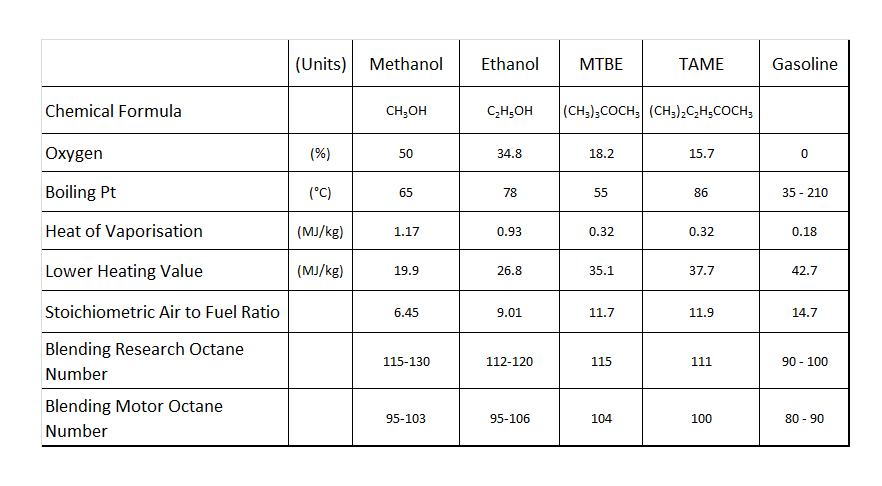 Fig. 1 - Common oxygenates
Fig. 1 - Common oxygenates
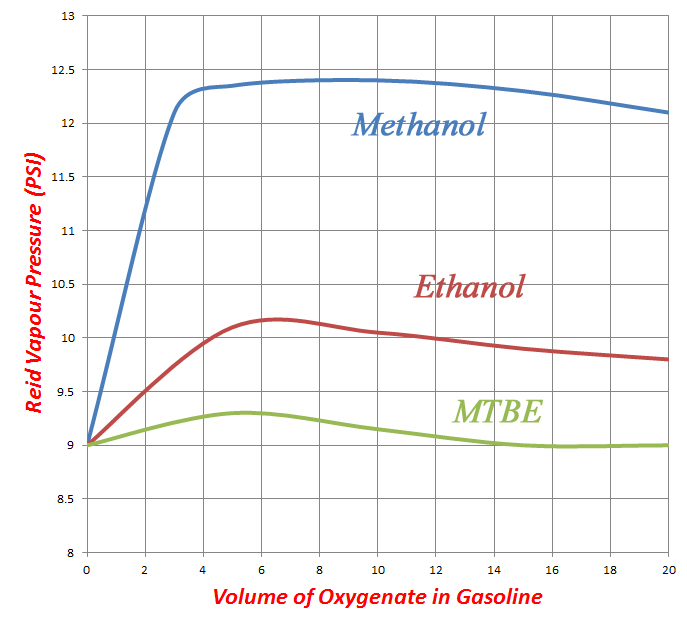 Fig. 2 - Effect on vapour pressure of key oxygenates
Fig. 2 - Effect on vapour pressure of key oxygenates
Written by John Coxon