The Formula One gearbox lube
 If the lubricant for a Formula One engine poses one set of challenges then that needed in its gearbox creates some that are wholly different. While the internals of a Formula One V8 engine are familiar, if slightly smaller than many may be used to, those of the gearbox are more akin to the movement of a Swiss watch. With gear widths sometimes of the order of 4-5 mm, and slipping in and out of use seamlessly, the analogy is reasonably close. But whereas a watch simply and relentlessly marks the passage of time, a Formula One gearbox needs to transmit prodigious amounts of torque at speeds up to 18,000 rpm. In doing so, there are only two criteria that predominate - friction and durability.
If the lubricant for a Formula One engine poses one set of challenges then that needed in its gearbox creates some that are wholly different. While the internals of a Formula One V8 engine are familiar, if slightly smaller than many may be used to, those of the gearbox are more akin to the movement of a Swiss watch. With gear widths sometimes of the order of 4-5 mm, and slipping in and out of use seamlessly, the analogy is reasonably close. But whereas a watch simply and relentlessly marks the passage of time, a Formula One gearbox needs to transmit prodigious amounts of torque at speeds up to 18,000 rpm. In doing so, there are only two criteria that predominate - friction and durability.
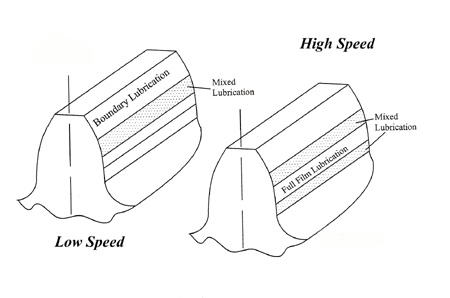
For engine lubricants the traditional choice is mineral oil or synthetic - PAOs (polyalphaolefins) and esters. In the case of transmissions, however, another family of synthetic products, polyglycols, may be commonly found. A member of the same family as used in some ethylene glycol, anti-freeze formulations, polyglycols are ideal for applications with high levels of the shearing action and boundary lubrication - or, if you like, when the relative motion between adjacent components, for instance in touching gear teeth, is small.
In these circumstances the coefficient of friction is very small, much smaller than other oils and possibly even half that of some synthetic products. Polyglycols also have high VIs (viscosity indices): that is, their viscosity changes less with temperature compared to other synthetic oils. The VI of a pure PAO oil might be 170-180 or so. For a polygylcol, the VI could be nearer 230, which is a major advantage at the extreme pressures reached in gear teeth.
While blends of other synthetics or mineral oils can achieve such high VIs using viscosity modifiers (VMs), the large molecules of these VMs can be readily chopped up under the shearing action of the gear teeth and quickly lose their effectiveness. In such cases therefore, the high-temperature viscosity of the oils can quickly fall away. The use of polyglycol technology can avoid such difficulties.
Unfortunately, the factor that makes polyglycols so attractive - that is, their affinity for the metal they are designed to protect - is the same reason for their undesirability. The highly polar oxygen atom, repeated regularly along the length of the chain, is readily attracted to positively charged metal surfaces. However, the hydrogen ions of water vapour are easily attracted to the same atoms, so oils based on polyglycols have a high affinity for water. And water, I don't need to remind you, is the enemy of anything ferrous. Polyglycols should never be used where water may be present and is the reason they can't be used in the engines. Since transmissions are generally sealed from any water vapour in the atmosphere, rust is rarely a problem inside most gearboxes.
Until recently, the other problem with polyglycols has been their incompatibility with other, more traditional hydrocarbon oils. So while many different types of synthetic products may be used in a Formula One gearbox, you can be almost certain that polyglycols will feature to a greater or lesser extent.
Fig. 1 - Lubrication zones in gear teeth
Written by John Coxon